Sodium butyrate alleviates R97-116 peptide-induced myasthenia
Por um escritor misterioso
Last updated 29 março 2025
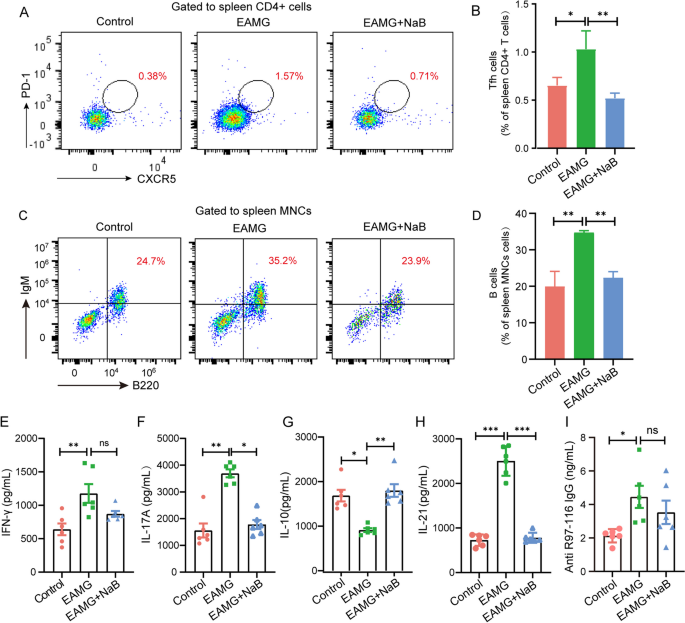
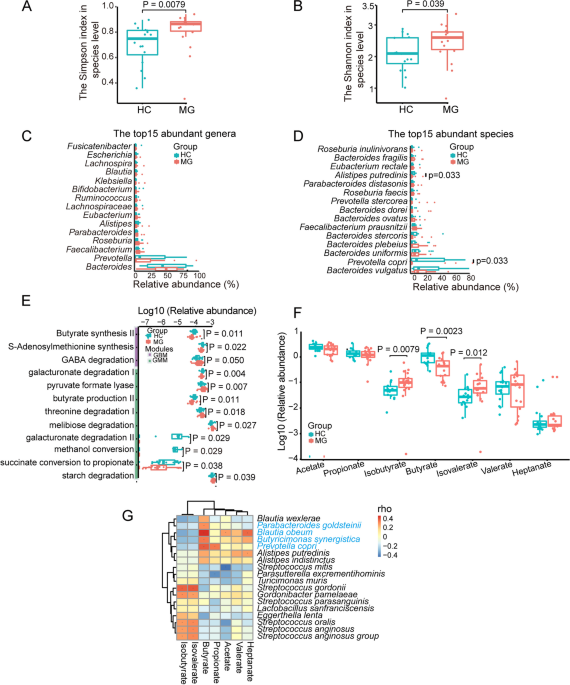
Fermented butyrate exhibits an anti-inflammatory response to maintain immune homeostasis within the gut. However, the effect and underlying mechanism of butyrate on myasthenia gravis (MG) remain unclear. The changes in the gut microbiota and fecal contents of SCFAs in MG patients were examined. R97-116 peptide was used to induce the experimental autoimmune myasthenia gravis (EAMG) mice and sodium butyrate (NaB) was gavaged to the EAMG mice. Gut microbiota, the frequency of Th1, Th17, Treg, Tfh, and B cells, the levels of IFN-γ, IL-17 A, IL-10, IL-21, and anti-R97-116 IgG, RNA-seq of total B cells in the spleen were explored by metagenomics, flow cytometry, ELISA, and transcriptomics. A significant reduction in SCFA-producing bacteria including Butyricimonas synergistica and functional modules including butyrate synthesis/production II was observed in MG patients and fecal SCFAs detection confirmed the increase. The EAMG mice were successfully constructed and NaB supplementation has changed the composition and function of the gut microbiota. The numbers of Th1, Th17, Tfh, and B cells were significantly increased while that of Treg cells was obviously decreased in EAMG mice compared with controls. Interestingly, NaB treatment has reduced the amounts of Th17, Tfh, and B cells but increased that of Treg cells. Accordingly, the levels of IL-17 A, IL-21, and IgG were increased while IL-10 was decreased in EAMG mice. However, NaB treatment reduced IL-17 A and IL-21 but increased that of IL-10. RNA-seq of B cells has revealed 4577 deferentially expressed genes (DEGs), in which 1218 DEGs were up-regulated while 3359 DEGs were down-regulated in NaB-treated EAMG mice. GO enrichment and KEGG pathway analysis unveiled that the function of these DEGs was mainly focused on immunoglobulin production, mitochondrial respiratory chain complex, ribosome, oxidative phosphorylation, and CNS diseases including amyotrophic lateral sclerosis. We have found that butyrate was significantly reduced in MG patients and NaB gavage could evidently improve MG symptoms in EAMG mice by changing the gut microbiota, regulating the immune response, and altering the gene expression and function of B cells, suggesting NaB might be a potential immunomodulatory supplement for MG drugs.
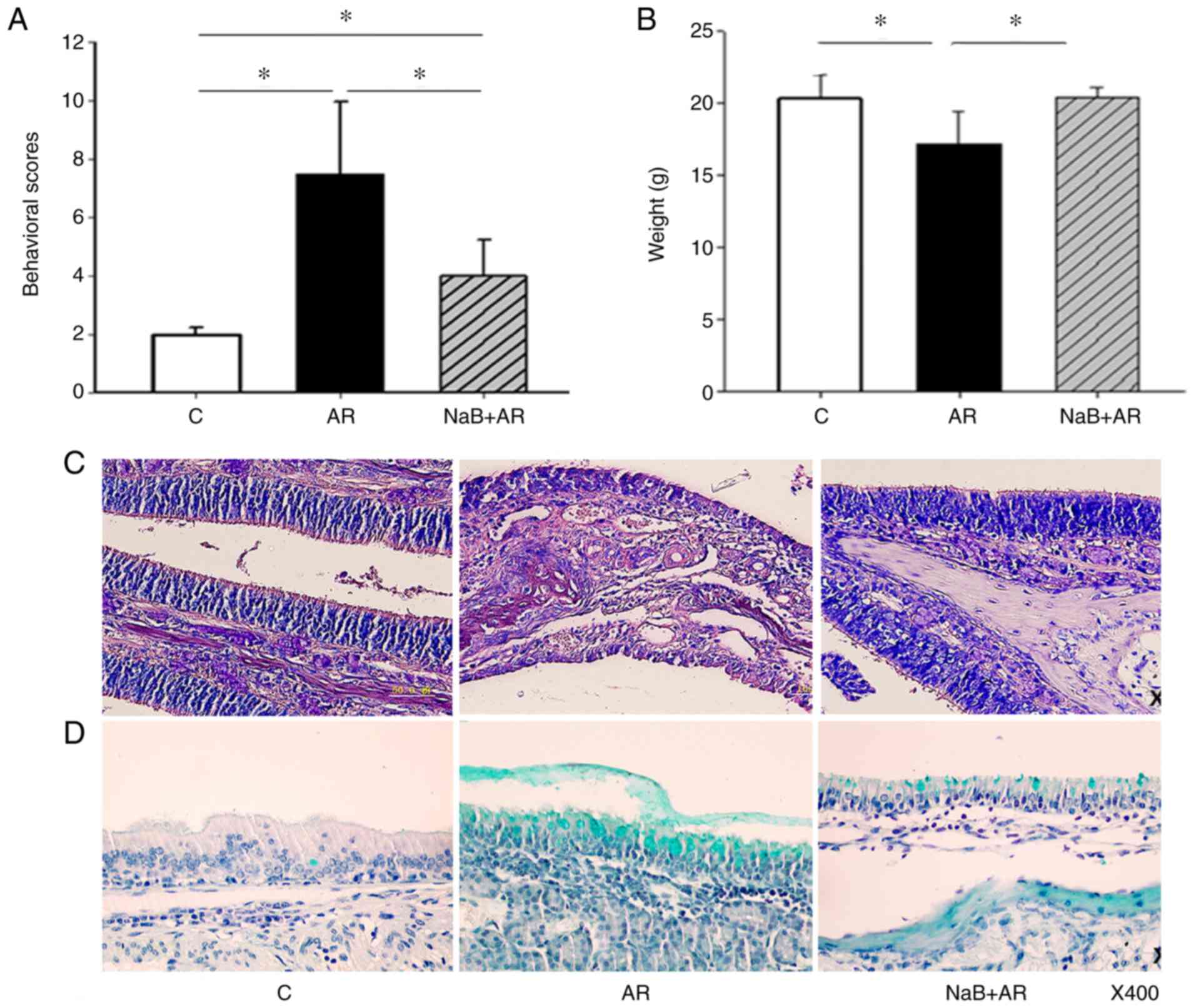
HDAC inhibitor sodium butyrate prevents allergic rhinitis and alters lncRNA and mRNA expression profiles in the nasal mucosa of mice
Sodium butyrate alleviates R97-116 peptide-induced myasthenia gravis in mice by improving the gut microbiota and modulating immune response, Journal of Inflammation

PDF) Sodium butyrate alleviates R97-116 peptide-induced myasthenia gravis in mice by improving the gut microbiota and modulating immune response
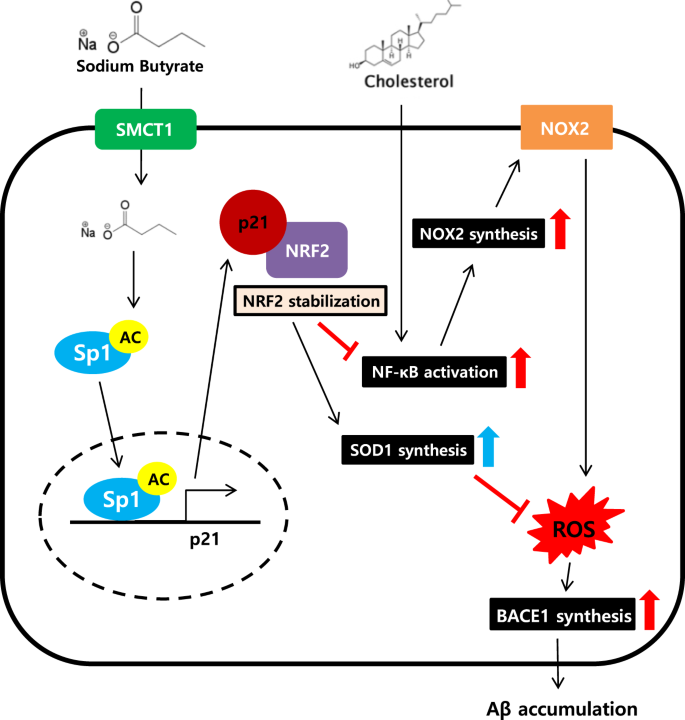
Sodium butyrate inhibits high cholesterol-induced neuronal amyloidogenesis by modulating NRF2 stabilization-mediated ROS levels: involvement of NOX2 and SOD1
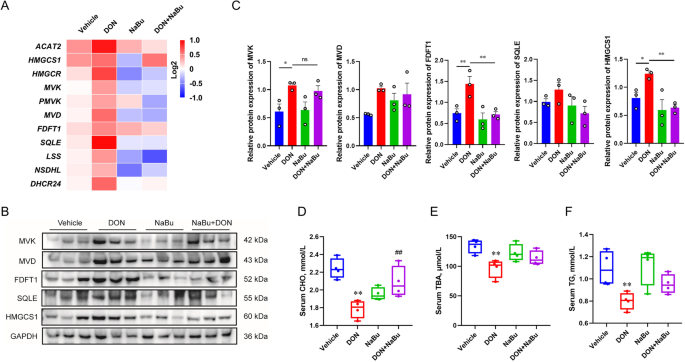
Sodium butyrate alleviates deoxynivalenol-induced hepatic cholesterol metabolic dysfunction via RORγ-mediated histone acetylation modification in weaning piglets, Journal of Animal Science and Biotechnology

PDF) Sodium butyrate alleviates R97-116 peptide-induced myasthenia gravis in mice by improving the gut microbiota and modulating immune response

Visualization of nucleotide substitutions in the (micro)transcriptome

Dietary butyrate treatment enhances healthy metabolites by longitudinal untargeted metabolomic analysis in amyotrophic lateral sclerosis mice
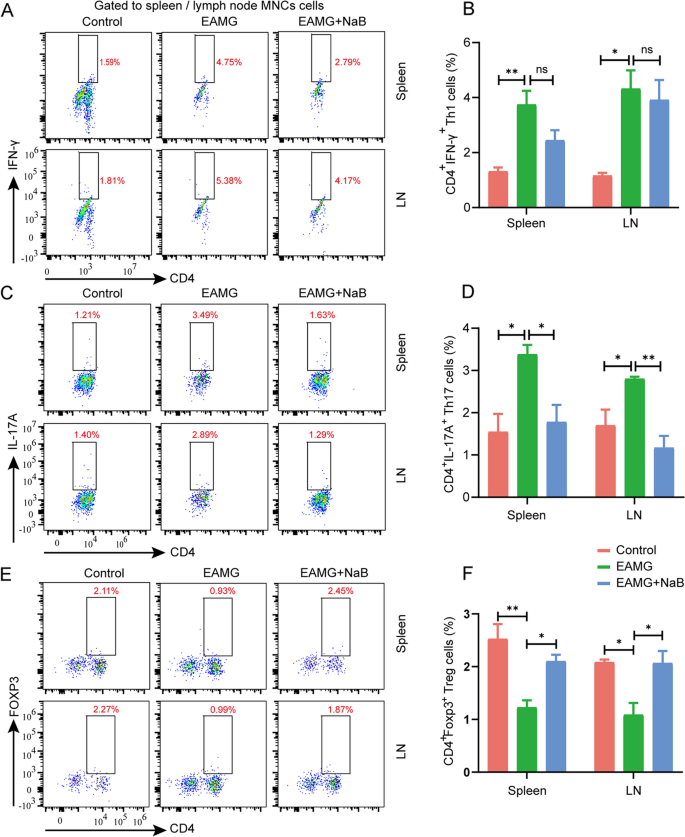
Sodium butyrate alleviates R97-116 peptide-induced myasthenia gravis in mice by improving the gut microbiota and modulating immune response, Journal of Inflammation
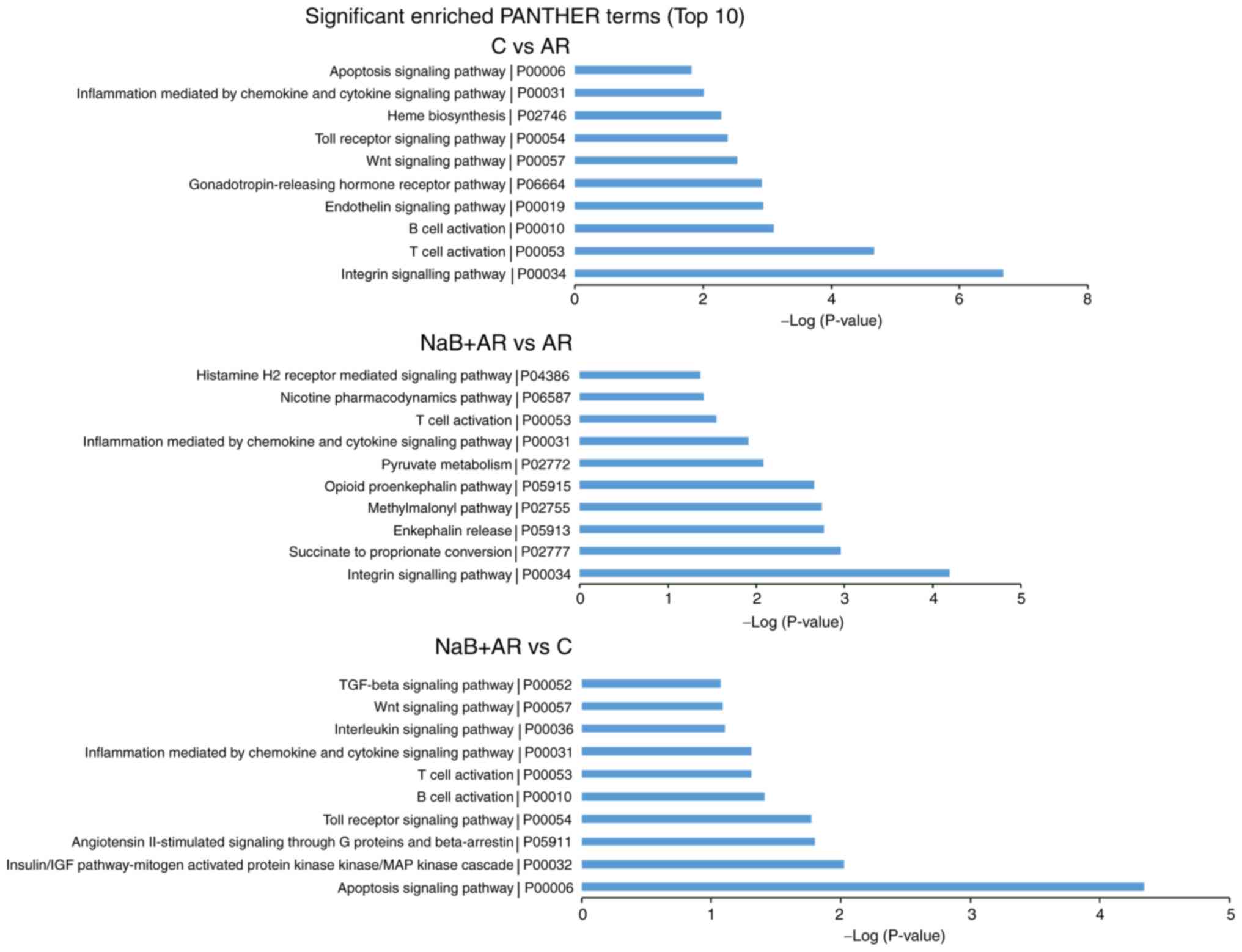
HDAC inhibitor sodium butyrate prevents allergic rhinitis and alters lncRNA and mRNA expression profiles in the nasal mucosa of mice

Visualization of nucleotide substitutions in the (micro)transcriptome
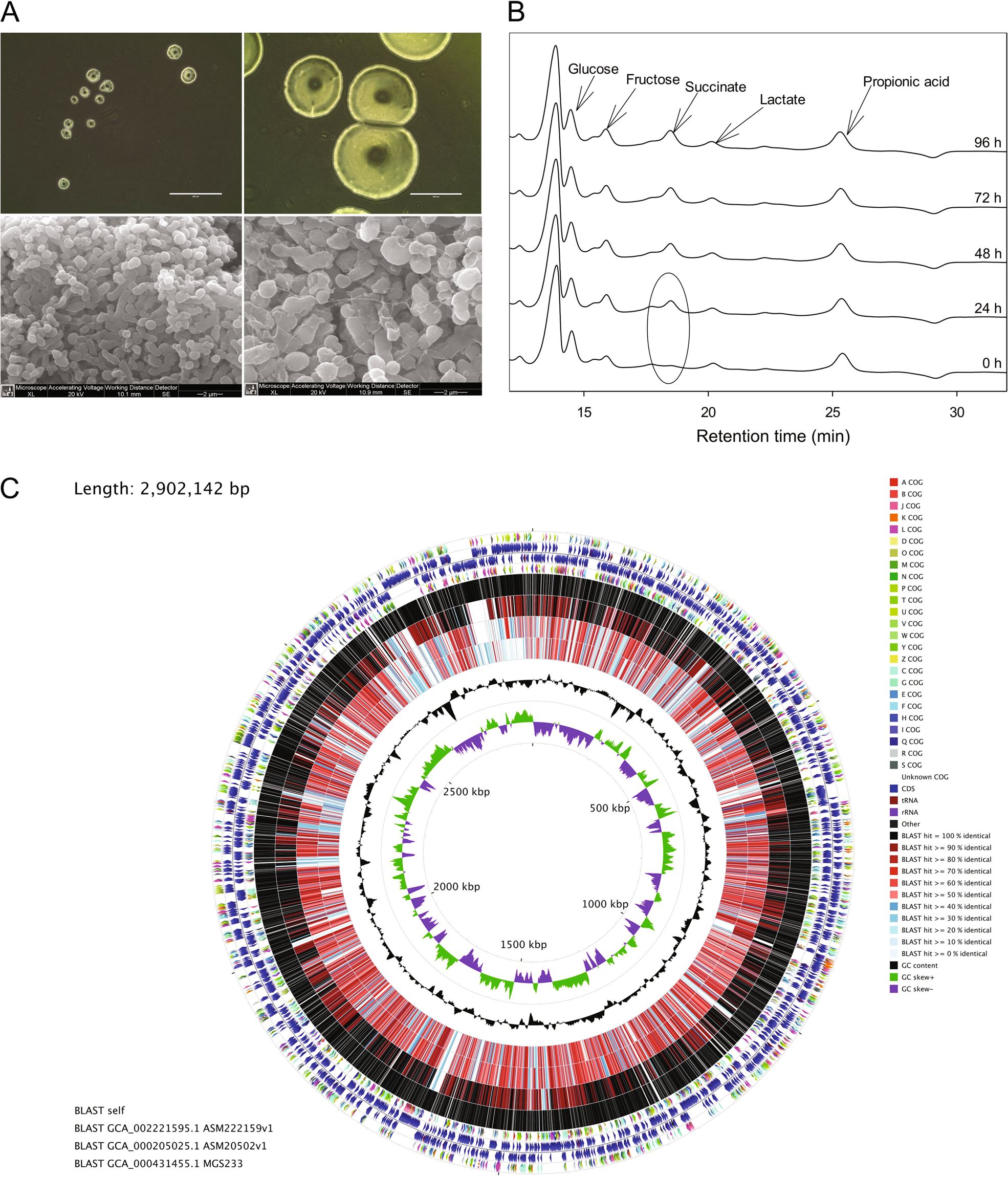
Defining the role of Parasutterella, a previously uncharacterized member of the core gut microbiota
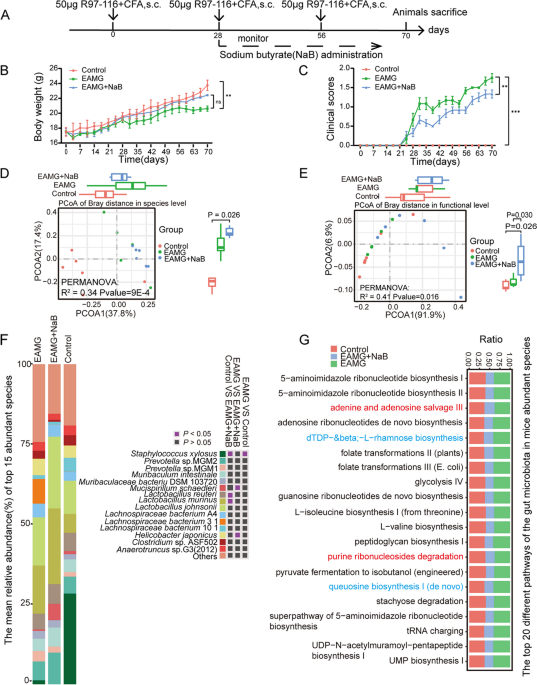
Sodium butyrate alleviates R97-116 peptide-induced myasthenia gravis in mice by improving the gut microbiota and modulating immune response, Journal of Inflammation

Endogenous murine microbiota member Faecalibaculum rodentium and its human homologue protect from intestinal tumour growth. - Abstract - Europe PMC
Recomendado para você
-
Omega Legends - Apps on Google Play29 março 2025
-
 Shotgun King: The Final Checkmate Free Download (v1.39c) « IGGGAMES29 março 2025
Shotgun King: The Final Checkmate Free Download (v1.39c) « IGGGAMES29 março 2025 -
 Fetal genome profiling at 5 weeks of gestation after noninvasive29 março 2025
Fetal genome profiling at 5 weeks of gestation after noninvasive29 março 2025 -
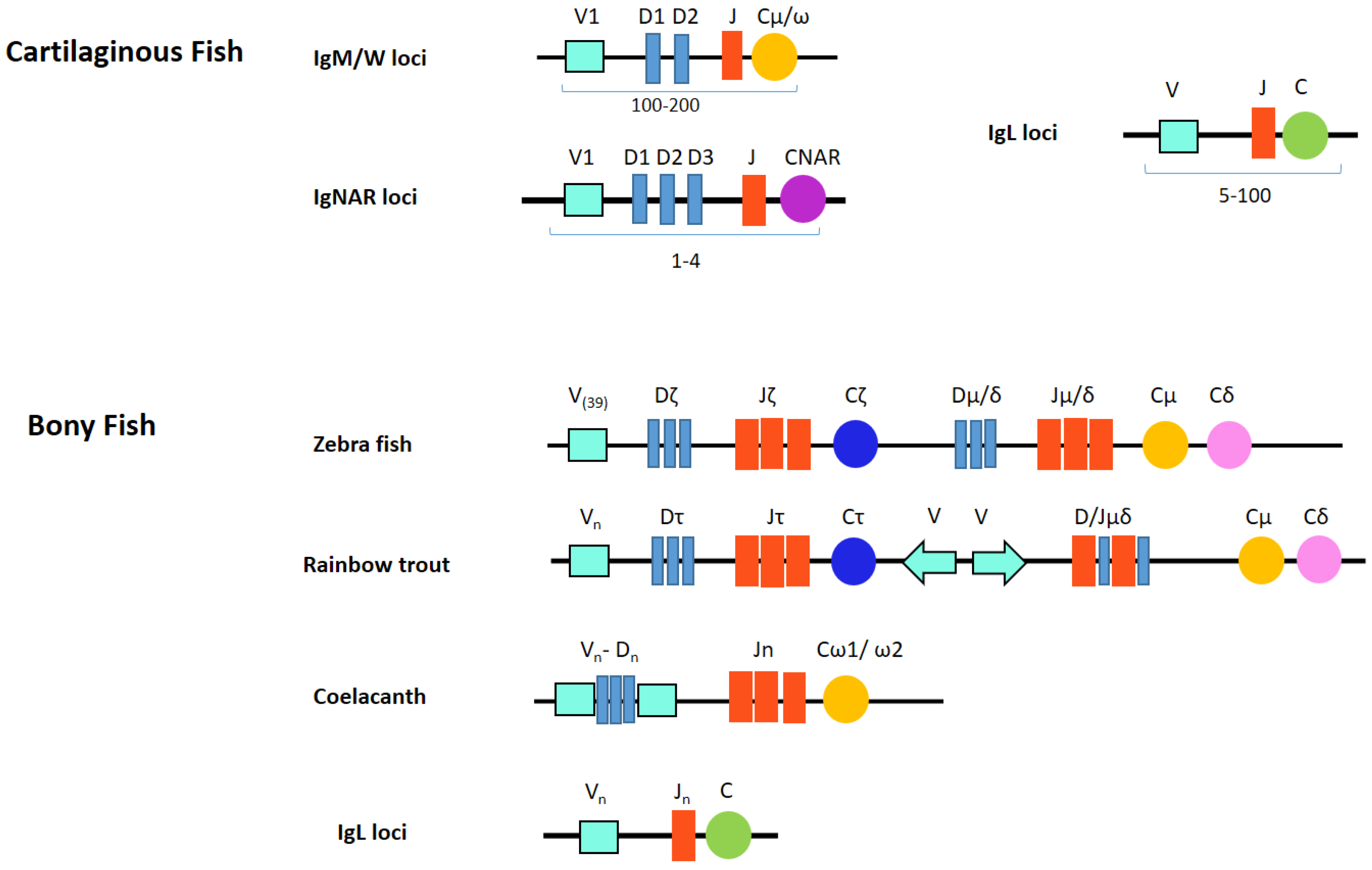 Biology, Free Full-Text29 março 2025
Biology, Free Full-Text29 março 2025 -
 Faisal ALASMARI, King Fahad Medical City, Riyadh29 março 2025
Faisal ALASMARI, King Fahad Medical City, Riyadh29 março 2025 -
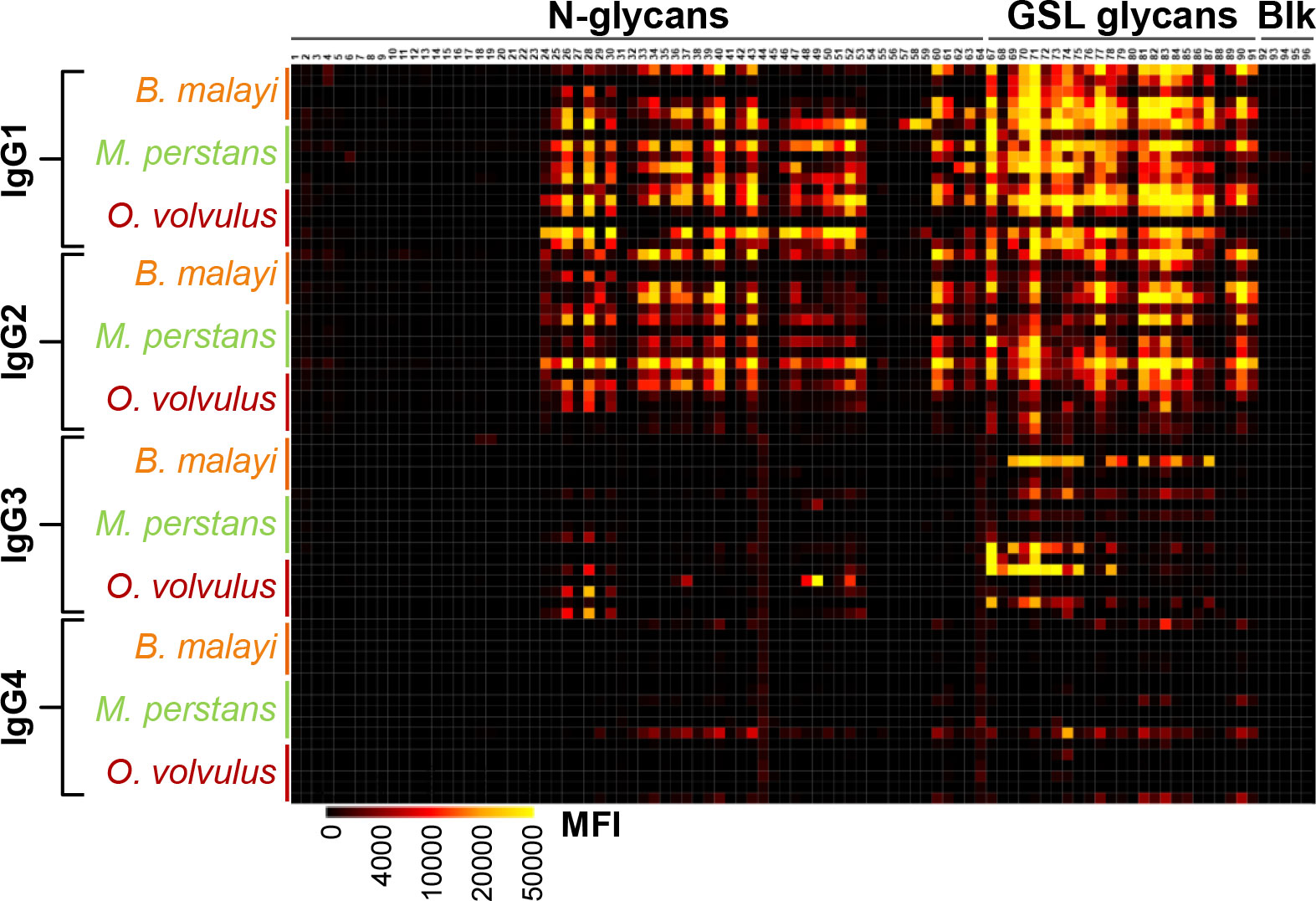 Frontiers Unraveling cross-reactivity of anti-glycan IgG29 março 2025
Frontiers Unraveling cross-reactivity of anti-glycan IgG29 março 2025 -
 Castle Clash, Character catalogue Wiki29 março 2025
Castle Clash, Character catalogue Wiki29 março 2025 -
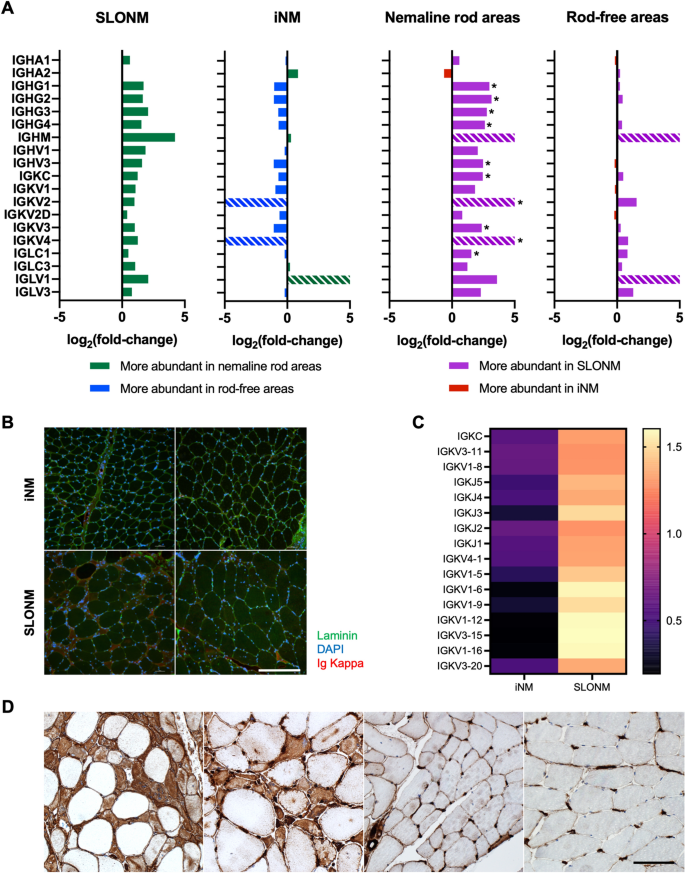 Molecular signatures of inherited and acquired sporadic late onset29 março 2025
Molecular signatures of inherited and acquired sporadic late onset29 março 2025 -
 Brainworx bx_opto - Electronic Delivery - Vintage King29 março 2025
Brainworx bx_opto - Electronic Delivery - Vintage King29 março 2025 -
 Shotgun King: The Final Checkmate by PUNKCAKE Délicieux 🥞, Benjamin Soulé29 março 2025
Shotgun King: The Final Checkmate by PUNKCAKE Délicieux 🥞, Benjamin Soulé29 março 2025
você pode gostar
-
 Live A Live JRPG / RPG - (SNES) English Translated – Retro Gamers US29 março 2025
Live A Live JRPG / RPG - (SNES) English Translated – Retro Gamers US29 março 2025 -
 Mahou Shoujo ni Akogarete – Comédia sobre garota virando vilã de sua heroína favorita ganha visual e janela de estreia - IntoxiAnime29 março 2025
Mahou Shoujo ni Akogarete – Comédia sobre garota virando vilã de sua heroína favorita ganha visual e janela de estreia - IntoxiAnime29 março 2025 -
 Ice Cream Roll Maker - Make Amazing Ice Cream Desserts29 março 2025
Ice Cream Roll Maker - Make Amazing Ice Cream Desserts29 março 2025 -
 Jogo De Bolas Profissional Sinuca / Snooker / Bilhar 50mm29 março 2025
Jogo De Bolas Profissional Sinuca / Snooker / Bilhar 50mm29 março 2025 -
 New Nerf N-Strike Elite Centurion Mega Sniper Blaster Toy Foam29 março 2025
New Nerf N-Strike Elite Centurion Mega Sniper Blaster Toy Foam29 março 2025 -
 So Not My Type #imjustakidchallenge Compilation #webtoon29 março 2025
So Not My Type #imjustakidchallenge Compilation #webtoon29 março 2025 -
 Radnički 1923 sezonu počinje protiv Novog Pazara – Ritam Grada – vesti Kragujevac29 março 2025
Radnički 1923 sezonu počinje protiv Novog Pazara – Ritam Grada – vesti Kragujevac29 março 2025 -
 RuPaul's Drag Race UK Series 3 / Characters - TV Tropes29 março 2025
RuPaul's Drag Race UK Series 3 / Characters - TV Tropes29 março 2025 -
 Download Roblox Shirt For Boy Template - Full Size PNG Image - PNGkit29 março 2025
Download Roblox Shirt For Boy Template - Full Size PNG Image - PNGkit29 março 2025 -
 Door Guardian, Lies Of P Wiki29 março 2025
Door Guardian, Lies Of P Wiki29 março 2025